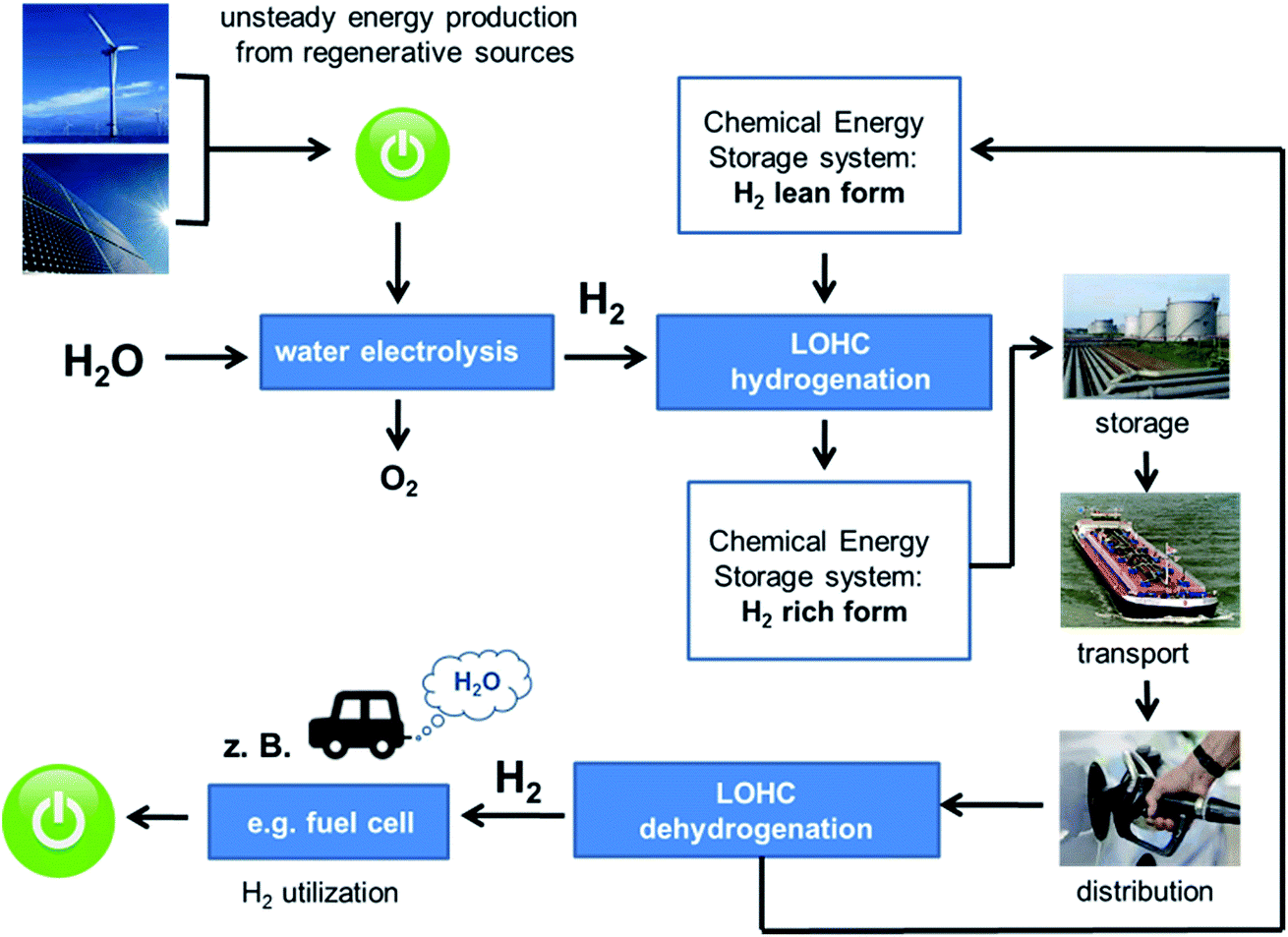
Energy storage and transport
If renewable energies are to be used to a larger extent than today, technologies for storage and transport of those energies are needed. Since wind- and solar energies are not produced “on demand” but rather are generated irregularly they have to be stored until they are needed. For mobile applications like cars transportable storage and release systems have to be developed.
One approach to store energy, for example from wind power, is the generation of hydrogen by electrolysis. Since free hydrogen is not easily stored, we aim at converting hydrogen to storable compounds. This can be achieved by hydrogenationg suitable substances. Those compounds are referred to as “Liquid Organic Hydrogen Carriers”, LOHC (Energy Environ. Sci. 2011, 4, 2767-2773. DOI: 10.1039/C1EE01454D).
One possible molecule that can serve as LOHC is perhydro-N-ethylcarbazole. (LOHC, loaded form). This compound is liquid at room temperature and not easily flamable. Its handling properties are similar to conventional diesel fuel. Perhydrocarbazol can be stored and transported by conventional means. The hydrogen is released by catalytic dehydrogenation. It can then be used in fuel cells to generate electricity. The discharged form of the LOHC, the ethylcarbazole, is then re-hydrogenated and can be used as energy carrier again.